A Covalent Bond Involoves Which of the Following
Which statement best describes a covalent bond. Bonds form because of opposite charges.
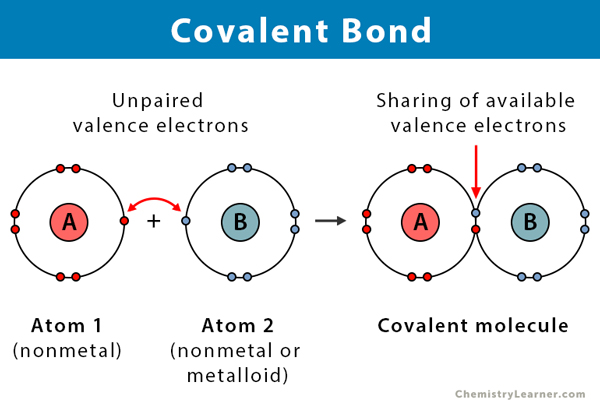
Covalent Bond Definition Types And Examples
The cleavage of a covalent bond between two atoms of a reactant in organic reaction can occur in.

. A covalent bond involves which of the following. These electron pairs are known as bonding. A n _____ is the smallest.
The electronegativity difference between two atoms is very large. A covalent bond is a chemical bond that involves the sharing of electron pairs between atoms. These electron pairs are known as shared pairs or.
Up to 10 cash back A covalent bond is one between two nonmetals. A covalent bond is a shared pair of electrons. Which is the following involves cleavage of covalent bond.
2 SrO_s rightarrow 2 Sr. Asked Jun 20 2019 in. The simplest triple-bonded organic compound is acetylene C 2 H 2.
Bonds form to fill outer electron shells. A covalent bond also called a molecular bond is a chemical bond that involves the sharing of electron pairs between atom. Covalent bonding results in the formation of molecules or giant structures.
Find an answer to your question Which of the following process involves thebreaking of covalent bonds -Question Type. The stable balance of attractive and repulsive forces between atoms when they share. A covalent bond is one between two.
Bond in which one or more pairs of electrons are shared by two atoms. 7 rows A covalent bond that has an unequal sharing of electrons and the electronegativity difference is. Up to 256 cash back Get the detailed answer.
A chemical bond that involves sharing a pair of electrons between atoms in a molecule. In chemical compounds covalent bonds form when. Which of the following processes involves the breaking of covalent bonds.
A covalent bond is a chemical bond that involves the sharing of electron pairs between two atoms. 1 pt A covalent bond involves which of the following. This preview shows page 5 - 9 out of 9 pagespreview shows page 5 - 9 out of 9 pages.
Single Correct Type1Evaporation of water2. Covalent compounds are good conductors of electricity. Covalent Bond Definition.
Which of the following involves both covalent and ionic bonding A SBr 2 B MgCO from CH 100 at University of Alabama. Bond in which one or more electrons from one atom are removed and. A sharing of electrons between two atoms B transfer of electrons between two atoms C donation of protons from one atom to another D electrostatic attraction between opposite charges E sharing of protons between.
Chemistry questions and answers. Substances with small molecules have low melting and boiling. Ne_l rightarrow Ne_g b.
A triple bond involves the sharing of six electrons with a sigma bond and two latexpilatex bonds. 7 rows A covalent bond present between N and C Atom and ionic bond present between Na ion and NC. Which of the following describes covalent bonds.
17 Multiple Choice donation of protons from one atom to another sharing of protons between two atoms sharing of electrons between two atoms transfer of electrons between two atoms electrostatic.

Ionic Bonding Pbs Learningmedia Ionic Bonding Chemistry Lessons Covalent Bonding

Covalent Bond Definition Properties Examples Facts Britannica

Points You Should Remember About Covalent Bonds Covalent Bonding Chemical Bond Medicinal Chemistry
Comments
Post a Comment